SOLVED: To determine the ammonia concentration in a sample of lake water; three samples are prepared. In sample A, 10.0 mL of lake water is mixed with 5 mL of phenol solution

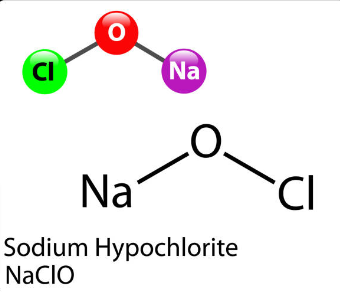
SOLVED: Sodium hydroxide reacts with chlorine gas to form sodium hypochlorite sodium chloride and water: bNaoH + ch QN C NaCl 3h,o Write the formula of sodium hypochlorite in the above blank












:max_bytes(150000):strip_icc()/make-your-own-disinfectant-solution-998274-V1-16d759206c054b1fb53410b90c57744c.jpg)