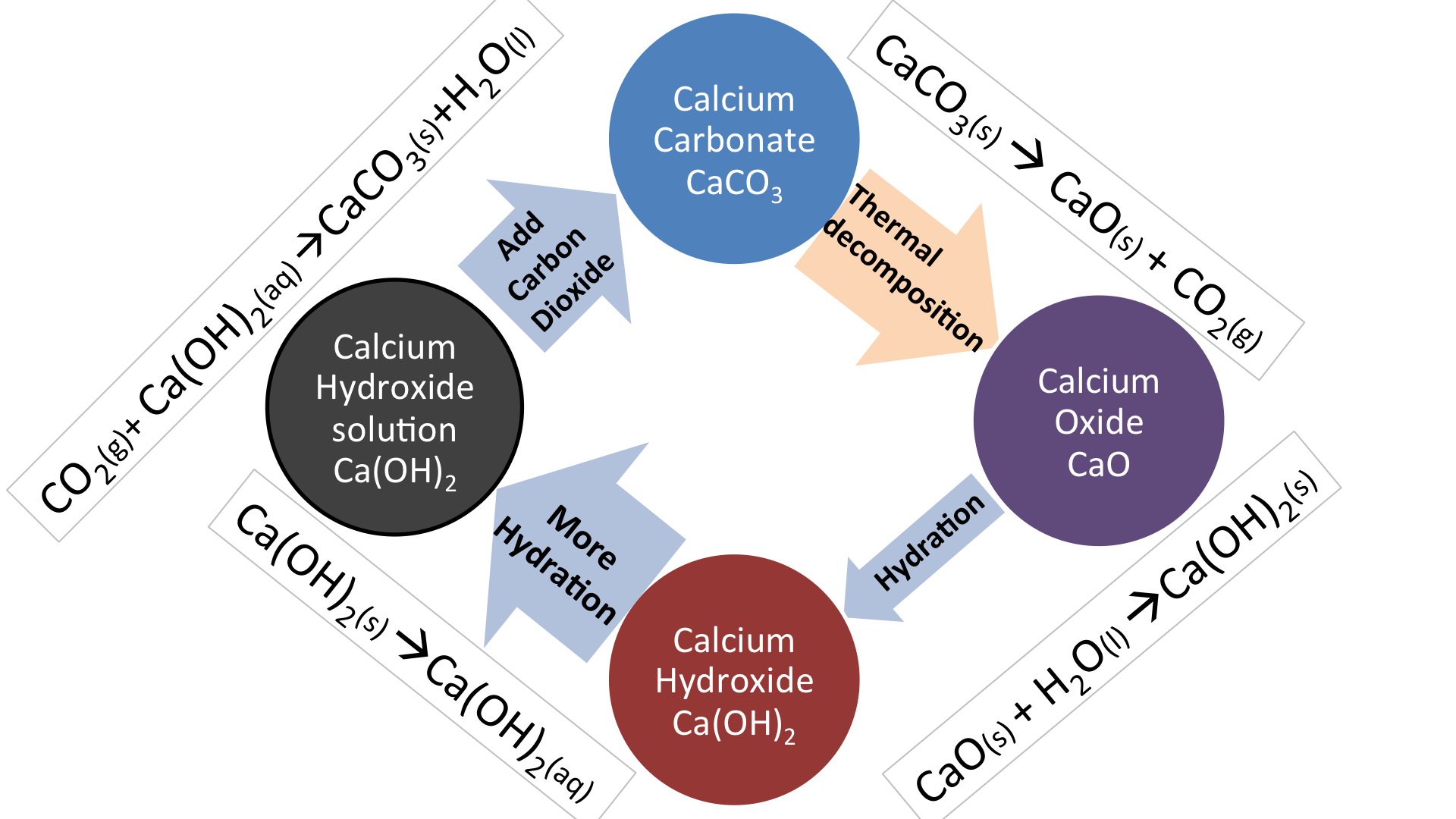
2.5 g of pure calcium carbonate, when strongly heated left a residue of 1.400 g. The evolved gas was found to occupy 624 mL at 27^@ C and 755 mm pressure. Calculate

Amazon.com: Schiff Calcium Carbonate Plus Magnesium with Vitamin D3 800 IU, Calcium Supplement, 90 ct (Pack of 3) : Health & Household
Calcium Carbonate Excipient | Uses, Suppliers, and Specifications | PharmaCentral | Materials and Knowledge Platform

Water Is the Key to Nonclassical Nucleation of Amorphous Calcium Carbonate | Journal of the American Chemical Society

SOLVED: How do I find the experimental Yield of CaCO3? when the mass of Calcium Chloride is 2.0 g, Sodium Carbonate is 2.5 g and the Filter paper is 4.3 g
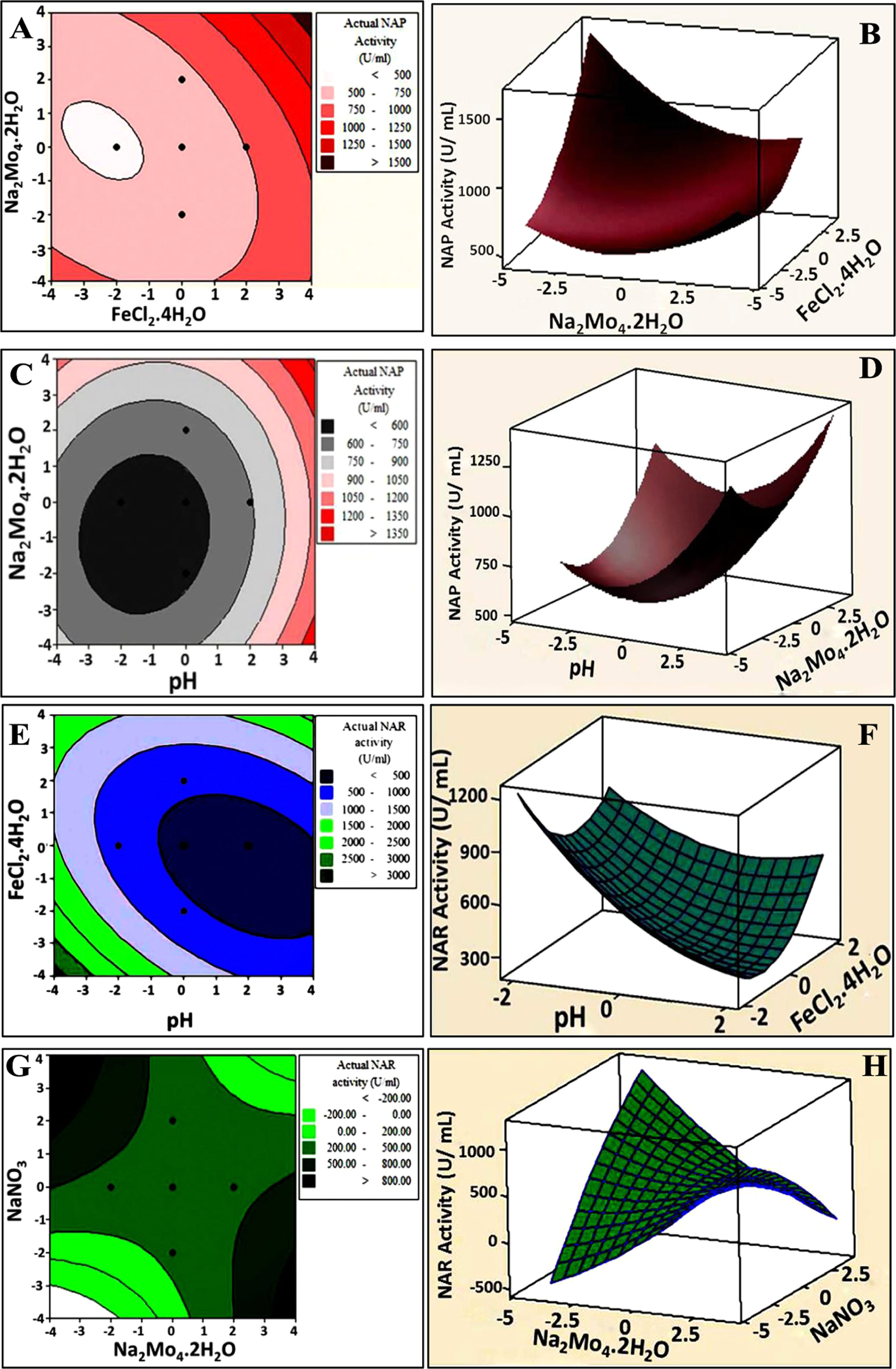
Aerobic and anaerobic removal of lead and mercury via calcium carbonate precipitation mediated by statistically optimized nitrate reductases | Scientific Reports

OneClass: Question 5. how many moles of oxygen are present in 300.0 g of calcium carbonate? the molar...

Density-dependent microbial calcium carbonate precipitation by drinking water bacteria via amino acid metabolism and biosorption - ScienceDirect